You are here
Ocean acidification and its effects
Oceans absorb a substantial proportion of the CO2 emitted into the atmosphere by human activities, with potentially negative effects on shell-forming organisms
At a glance
Oceans absorb a substantial proportion of the CO2 emitted into the atmosphere by human activities, with potentially negative effects on shell-forming organisms.
- Increasing CO2 in the atmosphere due to human activities not only affects the climate; it also has direct, chemical effects on ocean waters.
- The oceans have absorbed between a third and a half of the CO2 humans have released into the atmosphere since about 1850. This has slowed the rate of climate change.
- When CO2 dissolves in seawater, the water becomes more acidic. The acidity of the oceans has increased by 26 % since about 1850, a rate of change roughly 10 times faster than any time in the last 55 million years.
- Associated chemical reactions can make it difficult for marine calcifying organisms, such as coral and some plankton, to form shells and skeletons, and existing shells become vulnerable to dissolution.
- The extent to which calcifying organisms are already being affected by acidification is unclear, as this is a very new area of study. Limited evidence suggests that some organisms are more sensitive than others.
- The rate at which acidification occurs is a determining factor in the extent to which calcifying organisms will be able to adapt.
- The impacts of acidification will extend up the food chain to affect economic activities such as fisheries, aquaculture and tourism. Wherever there are marine calcifying organisms, there are risks from ocean acidification.
Main text
What is ocean acidification?
Human activities release CO2 into the atmosphere, which leads to atmospheric warming and climate change, as explained in Causes of climate change. Around a third to a half of the CO2 released by human activities is absorbed into the oceans. While this helps to reduce the rate of atmospheric warming and climate change, it also has a direct, chemical effect on seawater, which we call ocean acidification (Figure 1).
T2I3_Figure-1.gif
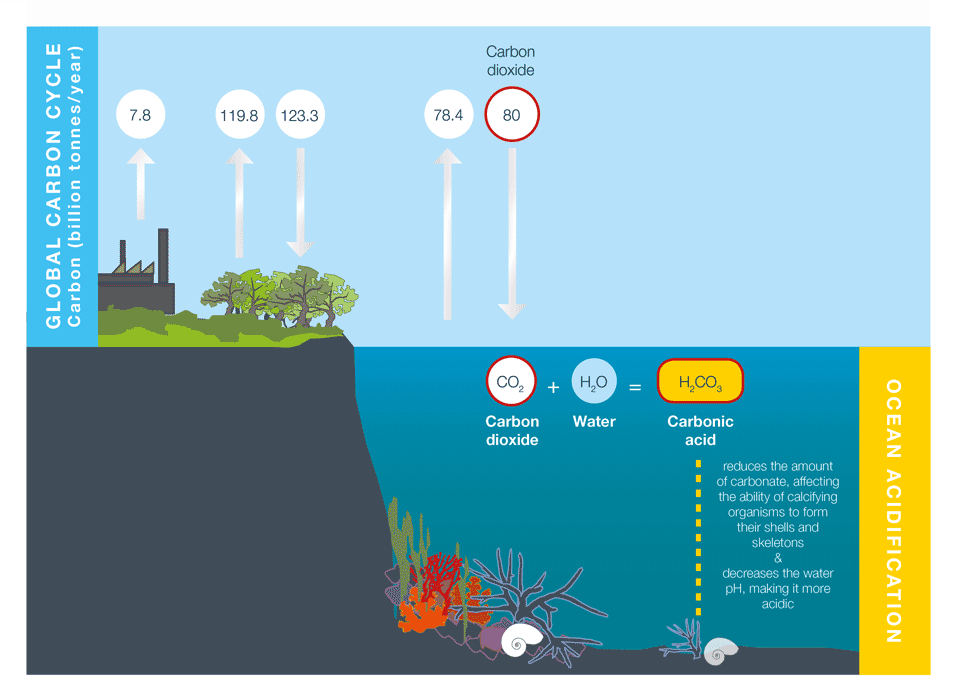
Box 1: What is pH?
Ocean acidification is often expressed in terms of the pH of seawater. pH is a measure of acidity or alkalinity. A pH below 7 is considered acidic, and a pH greater than 7 is considered alkaline, or basic.
Average ocean water pH is currently 8.1. The pH scale is logarithmic, so a one point change on the scale means a tenfold change in concentration.
T2I3_Figure B1.png

What are the observed changes?
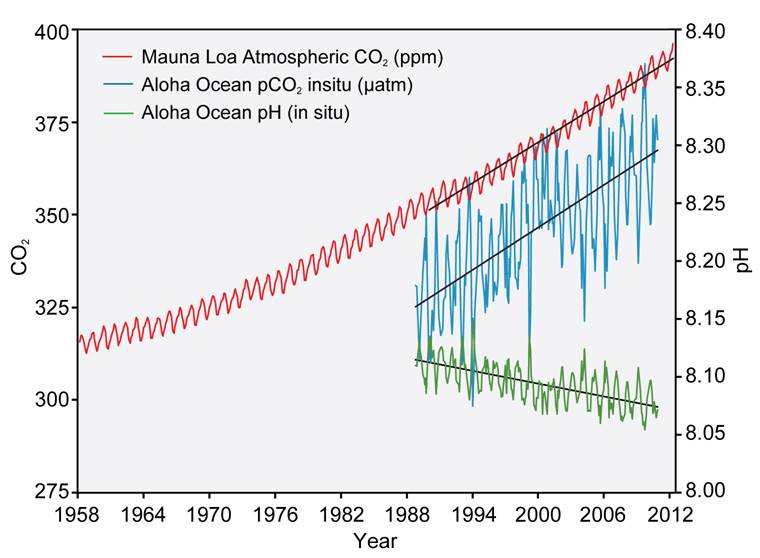
Since around 1850, the oceans have absorbed between a third and a half of the CO2 emitted to the atmosphere. As a result, the average pH of ocean surface waters has fallen by about 0.1 units, from 8.2 to 8.1 (Figure 2). This corresponds to a 26 % increase in ocean acidity, a rate of change roughly 10 times faster than any time in the last 55 million years.
T2I3_Figure-2.gif
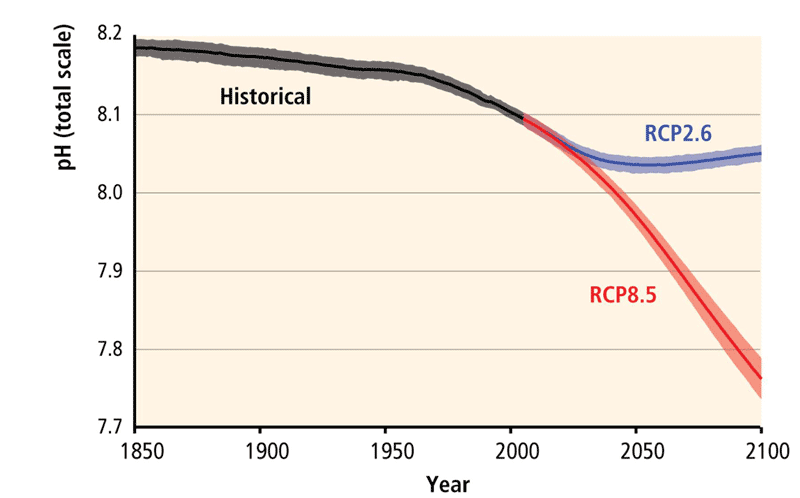
What can we expect in the future?
The degree of future ocean acidification will be very closely linked to future increases in atmospheric CO2 (Figure 3). If greenhouse gas emissions continue as they are doing at present (the RCP8.5 trajectory, see Causes of climate change), seawater could increase its acidity by 0.4 units (see Box 1) by the end of the century.
The acidification of the oceans will not be uniform worldwide. Polar seas, and upwelling regions, often found along the west coasts of continents, are expected to acidify faster than temperate or tropical regions. The pH will vary significantly depending on the ecosystem. In some parts of the Arctic the water is acidic enough to corrode some types of shells and in California occasional corrosive events have already occurred. Most surface waters will be continually corrosive within decades.
T2I3_Figure 3.jpg
What are the effects of ocean acidification on marine organisms and ecosystems?
Ocean acidification reduces the amount of carbonate, a key building block in seawater. This makes it more difficult for marine organisms, such as coral and some plankton, to form their shells and skeletons, and existing shells may begin to dissolve.
The present-day pH of seawater is highly variable, and a single organism can cope with fluctuations of different pH levels during its lifetime. The problem with ocean acidification is the sustained nature of the change, as the risk comes from the lifetime exposure to lower pH levels. The rapid pace of acidification will influence the extent to which calcifying organisms will be able to adapt.
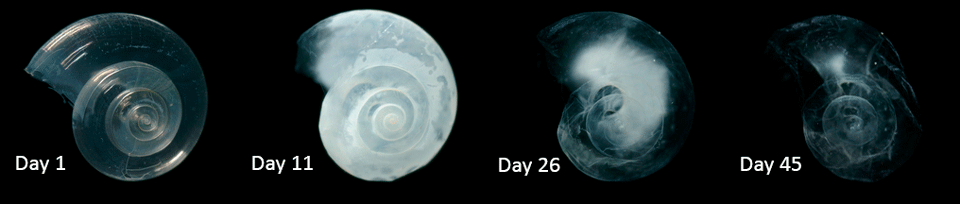
The impacts of ocean acidification are not uniform across all species. Some algae and seagrass may benefit from higher CO2 concentrations in the ocean, as they may increase their photosynthetic and growth rates. However, a more acidic environment will harm other marine species such as molluscs, corals and some varieties of plankton (Figure 4). The shells and skeletons of these animals may become less dense or strong. In the case of coral reefs this may make them more vulnerable to storm damage and slow the recovery rate.
T2I3_Figure-4 low res.gif
Marine organisms could also experience changes in growth, development, abundance, and survival in response to ocean acidification (Figure 5). Most species seem to be more vulnerable in their early life stages. Juvenile fish for example, may have trouble locating suitable habitat to live.
Despite the different responses within and between marine groups, positive or negative, research suggests that ocean acidification will be a driver for substantial changes in ocean ecosystems this century. These changes may be made worse by the combined effect with other emerging climate-related hazards, such as the decrease of ocean oxygen levels – a condition known as ocean deoxygenation –that is already affecting marine life in some regions (Long et al. 2016).
T2I3_Figure 5.jpg

What are the effects on human societies?
Changes in marine ecosystems will have consequences for human societies, which depend on the goods and services these ecosystems provide. The implications for society could include substantial revenue declines, loss of employment and livelihoods, and other indirect economic costs.
Socioeconomic impacts associated with the decline of the following ecosystem services are expected:
- Food: Ocean acidification has the potential to affect food security. Commercially and ecologically important marine species will be impacted, although they may respond in different ways. Molluscs such as oysters and mussels are among the most sensitive groups. By 2100, the global annual costs of mollusc loss from ocean acidification could be over US$100 billion for a business-as-usual (RCP8.5) CO2 emissions pathway.
- Coastal protection: Marine ecosystems such as coral reefs protect shorelines from the destructive action of storm surges and cyclones, sheltering the only habitable land for several island nations. This protective function of reefs prevents loss of life, property damage, and erosion, and has been valued at US$9 billion per year.
- Tourism: This industry could be severely affected by the impacts of ocean acidification on marine ecosystems (e.g. coral reefs). In Australia, the Great Barrier Reef Marine Park attracts about 1.9 million visits each year and generates more than A$5.4 billion to the Australian economy.
- Carbon storage and climate regulation: The capacity of the ocean to absorb CO2 decreases as ocean acidification increases. More acidic oceans are less effective in moderating climate change.
What can coastal decision makers do?
While reducing global greenhouse gas emissions (mitigation) is the ultimate solution to ocean acidification, undertaking some challenging decisions and actions can help us prepare for the adverse effects of ocean acidification. This is the adaptation approach.
At the local level, the following policy and management options can help to minimise the adverse effects of other local stressors and, as a result, help marine ecosystems to cope better with changing environmental conditions.
- Improvements in water quality: Monitoring and regulating localised sources of acidification from runoff and pollutants such as fertilisers.
- Development of sustainable fisheries management practices: Regulating catches to reduce overfishing and creating long-term bycatch[1] reduction plans.
- Implementation of new technologies: Different techniques can be applied depending on the industry. For example, in the aquaculture industry, new forecasting systems have been developed to account for seasonal upwellings that bring low pH seawaters to the ocean surface and cause massive shellfish die-offs.
- Sustainable management of habitats: Increasing coastal protection, reducing sediment loading and applying marine spatial planning.
- Establishment and maintenance of Marine Protected Areas: Protecting highly vulnerable and endangered marine ecosystems.
[1] Bycatch: a fish or other marine species that is caught unintentionally. Bycatch is either of a different species, the wrong sex, or is undersized or juvenile individuals of the target species.
Further information
The NOAA Ocean Acidification Program (US) provides useful and up to date information: http://oceanacidification.noaa.gov/Home.aspx (accessed 3 June 2016).
In Australia, scientist from the Australian Institute of Marine Sciences (AIMS) are researching the effects of ocean acidification on coral reef organisms and ecosystems: http://www.aims.gov.au/research/climate-change/ocean-acidification (accessed 3 June 2016).
This video developed by the Alliance for Climate Education provides an easy introduction to the topic: https://www.youtube.com/watch?v=Wo-bHt1bOsw.
However, if you are looking for more in-depth information, try watching this video developed by the World Bank Group's Open Learning Campus : https://www.youtube.com/watch?v=E39PMzDBtrU&feature=youtu.be&app=desktop.
Source material
Billé, R., and Coauthors, 2013: Taking action against ocean acidification: a review of management and policy options. Environmental management, 52(4), 761-779.
Brown, C. J., M.I. Saunders, H.P. Possingham, and A.J. Richardson, 2013: Managing for interactions between local and global stressors of ecosystems. PLoS One, 8(6).
Feely, R.A., C.L. Sabine, and V.J. Fabry, 2006: Carbon Dioxide and our Ocean Legacy. Accessed 17 May 2016. [Available online at http://www.pmel.noaa.gov/pubs/PDF/feel2899/feel2899.pdf].
Gattuso, J.P., P.G. Brewer, O. Hoegh-Guldberg, J.A. Kleypas, H.O. Pörtner, and D.N. Schmidt, 2014: Cross-chapter box on ocean acidification. In: Climate Change 2014: Impacts, Adaptation, and Vulnerability. Part A: Global and Sectoral Aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel of Climate Change, Field, C.B., V.R. Barros, D.J. Dokken, K.J. Mach, M.D. Mastrandrea, T.E. Bilir, M. Chatterjee, K.L. Ebi, Y.O. Estrada, R.C. Genova, B. Girma, E.S. Kissel, A.N. Levy, S. MacCracken, P.R. Mastrandrea, and L.L. White, Eds, World Meteorological Organization, Geneva, Switzerland, pp. 131-133. Accessed 17 May 2016. [Available online at https://www.ipcc.ch/pdf/assessment-report/ar5/wg2/WGIIAR5-IntegrationBrochure_FINAL.pdf].
IGBP, IOC, SCOR, 2013: Ocean Acidification Summary for Policymakers–Third Symposium on the Ocean in a High-CO2 World. International Geosphere-Biosphere Programme. Stockolm, Sweden. Accessed 17 May 2016. [Available online at http://www.igbp.net/download/18.30566fc6142425d6c91140a/1385975160621/OA_spm2-FULL-lorez.pdf].
IPCC, 2013: Summary for Policy Makers. In: Climate Change 2013: The Physical Science Basis. Contribution of Working Group I of the Fifth Assessment Report of the Intergovernmental Panel on Climate Change, Stocker, T.F., D. Qin, G.-K. Plattner, M.Tignor, S.K. Allen, J. Boschung, A. Nauels, Y. Xia, V. Bex and P.M. Midgley , Eds, Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA. Accessed 17 May 2016 [Available online at https://www.ipcc.ch/pdf/assessment-report/ar5/wg1/WG1AR5_SPM_FINAL.pdf].
Kroeker, K.J., R.L. Kordas, R. Crim, I.E. Hendriks, L. Ramajo, G.S. Singh, C.M. Duarte, and J.P. Gattuso, 2013: Impacts of ocean acidification on marine organisms: quantifying sensitivities and interaction with warming. Global Change Biology, 19(6), 1884-1896.
Long, M.C., C. Deutsch, and T. Ito, 2016: Finding forced trends in oceanic oxygen. Global Biogeochemical Cycles, 30(2), 381-397.
Narita, D., K. Rehdanz, and R.S. Tol, 2012: Economic costs of ocean acidification: a look into the impacts on global shellfish production. Climatic Change, 113(3-4), 1049-1063.
Noonan, S.H., and K.E. Fabricius, 2015: Ocean acidification affects productivity but not the severity of thermal bleaching in some tropical corals. ICES Journal of Marine Science: Journal du Conseil, fsv127. Accessed 17 May 2016. [Available online at http://icesjms.oxfordjournals.org/content/73/3/715.full.pdf+html].
Secretariat of the Convention of Biological Diversity, 2014: An Updated Synthesis of the Impacts of Ocean Acidification on Marine Biodiversity, Hennige,S., J.M. Roberts, and P. Williamson, Eds., Montreal, Technical Series No. 75, 99pp.
Turley, C., and J.-P Gattuso, 2012: Future biological and ecosystem impacts of ocean acidification and their socioeconomic-policy implications. Current Opinion in Environmental Sustainability, 4(3), 278-286.
Wittmann, A.C., and H.-O. Pörtner, 2013: Sensitivities of extant animal taxa to ocean acidification. Nature Climate Change, 3(11), 995-1001.
Wong, P.P., I.J. Losada, J.-P. Gattuso, J. Hinkel, A. Khattabi, K.L. McInnes, Y. Saito, and A. Sallenger, 2014: Coastal systems and low-lying areas. In: Climate Change 2014: Impacts, Adaptation, and Vulnerability. Part A: Global and Sectoral Aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change, Field, C.B., V.R. Barros, D.J. Dokken, K.J. Mach, M.D. Mastrandrea, T.E. Bilir, M. Chatterjee, K.L. Ebi, Y.O. Estrada, R.C. Genova, B. Girma, E.S. Kissel, A.N. Levy, S. MacCracken, P.R. Mastrandrea, and L.L. White, Eds, Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, pp. 361-409. Accessed 17 May 2016. [Available online at https://www.ipcc.ch/pdf/assessment-report/ar5/wg2/WGIIAR5-Chap5_FINAL.pdf].




